|
Electrolyzed
Water
Technology -
Electro-Chemical Activation
|

|

Electrolyzed water systems and electrolyzed water
Electro-Chemical Activation (ECA) technologies -
The most
advanced ECA Equipment available.
Our equipment is uniquely design to produce
Anolyte and Catholyte solutions with predictable and carefully
controlled properties.
Regardless
of the application, Anolyte at a neutral pH kills bacteria,
pathogens,
germs,
viruses, mold,
mildew,
fungi, and
spores on contact or within seconds.
Anolyte produced from our equipment has been
shown to be as much as
100 times
more effective than bleach at
eliminating bacteria.
|
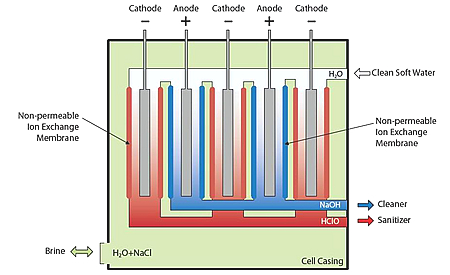
What are ECA Solutions?
Anolyte is a strong
oxidizing solution formed from naturally occurring elements
that kills unwanted microorganisms and pathogens.
Anolyte attacks an array of microbial organisms including
bacteria,
viruses, fungi, and
microbial spores.
Catholyte is
a mildly alkaline anti-oxidizing solution ideal for use as
degreaser, cleaner, and detergent.
Applications
- Disaster
clean-up and Sanitation (including mold remediation)
- Healthcare
û Medical, Dental, Surgical, Emergency Room,
Laboratory and Blood Bank applications
- Food
Services û Restaurants, Cafeterias and other public
eating
facilities
- Travel
Industry û Planes, Buses, Cruise Ships and Rental Cars
- Recreational
Facilities such as Gymnasiums, Health Clubs and
Bowling Alleys
- Restrooms,
Floors, Kitchens and Appliances
- Schools,
Daycare Facilities and Nurseries
- The Oil and
Gas Industry
- Hospitality
Industry
- Veterinary
Practices
- Many, Many
More!
Regulatory
Approval
|

|
U.S.
Department of Agriculture, Food Safety Inspection
Service (FSIS), approved for use on federally inspected
meat, poultry, or egg products |
|

|
U.S.
Food and Drug Administration (FDA) approved for use in
Dentistry. |
|

|
U.S.
Environmental Protection Agency (EPA) approved for the
disinfection of hard surfaces in medical, dental, and
veterinary applications. |
|

|
U.S.
Environmental Protection Agency (EPA) approved as a
biocide for oil and gas production. |
|

|
U.S.
Food and Drug Administration (FDA) approved for use in
meatpacking and processing plants as a replacement for
chlorine solutions. |
|
|
Pathogens
Eliminated
|
Adenovirus |
Glanders |
Noroviruses |
|
Anthrax |
H.
pylori |
Plague |
|
Avium-intracellltlare |
Haemophilus
influenzae |
Poliovirus |
|
Bacillus
anthracis |
Hepatitis
B |
Proteus |
|
Bacteria |
Hepatitis
C |
Proteus
mirabilis |
|
Botrytis
cinerea |
HIV |
Pseudomonas
aeruginosa |
|
Botulinum
toxin |
HIV-l |
Pseudomonas
fluorescens |
|
Brucellosis |
Klebsiella
Pneumonia |
S.
dublin |
|
Campylobacter |
Listeria
monocytogenes |
S.
haemolyticus |
|
Candida
albicans |
M.
avium |
S.
hominis |
|
Candida
albincans |
M.
chelonae |
S.
pullorum |
|
Coliphages |
M.
chelonei |
S.
saprophyticus |
|
Corynebacterium
amycolatum |
M.
luteus |
Salmonella
enterica |
|
Dermatomycosis |
M.
smegmatis |
Serritia
marcescens |
|
E.
coli |
M.
xenopi |
Staphlococcus
aureus |
|
Entamoeba
histolytica |
Mold |
Streptococcus
faecalis |
|
Enterobacter
aerogenes |
Monilinia
jructicola |
Streptococcus
pyogenes |
|
Enterobacter
cloaceae |
MRSA |
Sulfate
Reducing bacteria |
|
Enterococcus |
Mycobacterium |
Thermo-tolerant
coliform |
|
Enterococcus
faecalis |
Mycobacterium
tuberculosis |
Tularemia |
|
Fungi |
Methysillin
Resistant spores |
Yeast |
|
Aspergillus
niger |
General
coliform bacteria |
Vancomycin
Resistant spores of H. pylori |
|
|
of
Bacillus subtilis var niger |
|
|
|
|
|
|
|